NAME Biomarkatzaileak identifikatzeko eta kuantifikatzeko plataforma.
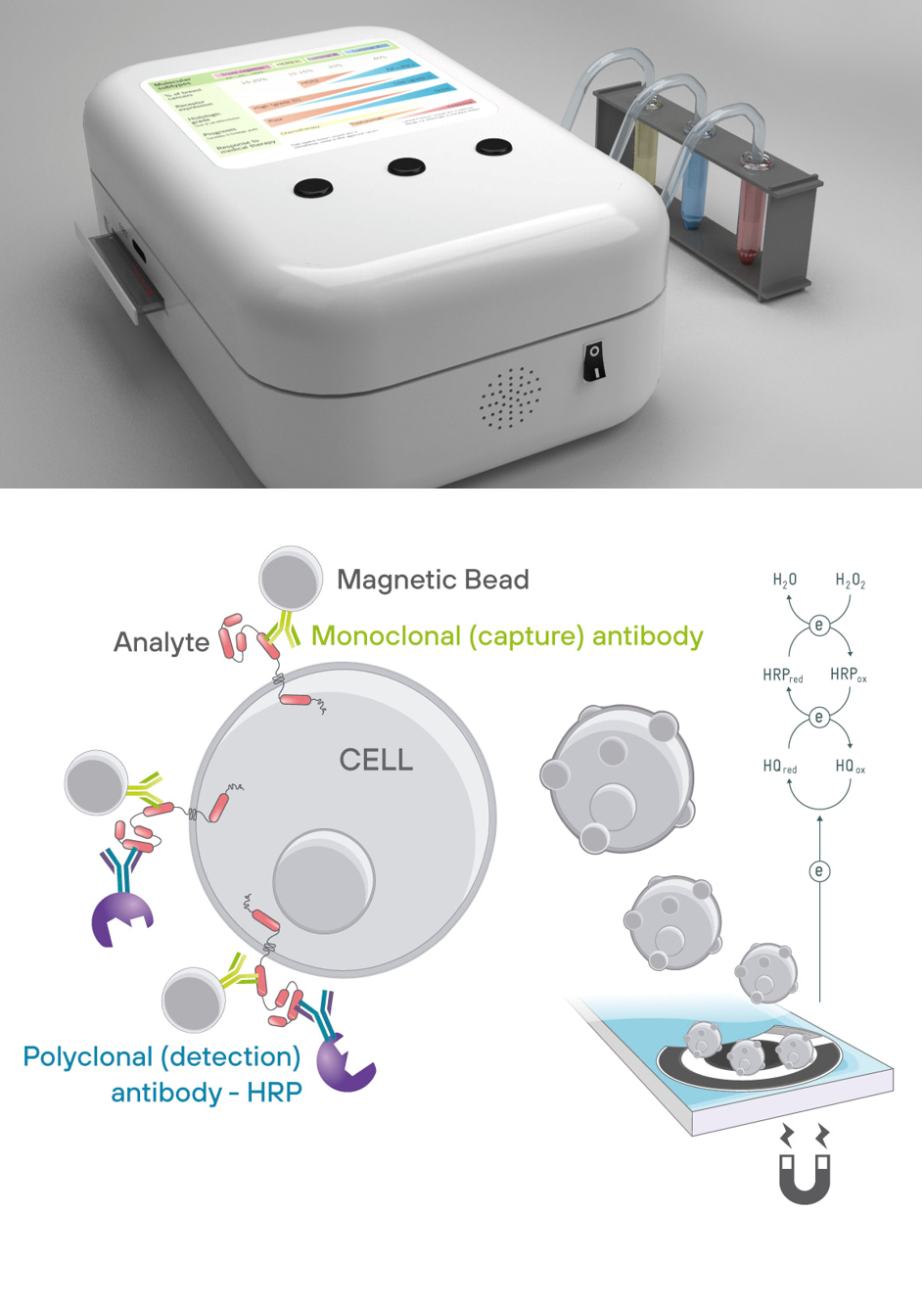
Mahaigaineko ekipamendua da, eta honela funtzionatzen du: - Sistemak automatikoki kargatzen ditu fluidoak eta lagina zirkuituan, erabiltzaileak agindutako prozesu-errezetari jarraituz. - Irabiatze-ganbera. Beharrezko produktuak, laginaren partikula magnetikoak eta antigorputz etiketatuak barne, sekuentzialki iristen dira immunokonplexua eratzen den barrunbera. - Neurketa-ganbera. Immunosaiakuntza behar bezala eratu ondoren, partikula magnetikoak ganberara bideratzen dira. Han, txip elektrokimikoaren azpian dagoen iman iraunkor batek atzematen ditu magnetikoki. Txiparen goialdean atzemandako partikula magnetikoek erreakzio bat sortzen dute, lagineko analito-kontzentrazioarekiko proportzionala. - Lagina kargatu eta 30 minutura lortzen da emaitza. Serum-lagin errealetan detekzio-sentsibilitate handiz (10 pg/ml ingurukoa) baliozkotutako teknologia
MOST OUTSTANDING EQUIPMENT AND COMPONENTS
-
Clean room
Clean room with photolithography, layer deposition and micro/nano structuring for the manufacture of microfluidic components.
-
Microfluidic platform for electrochemical detection
Microfluidic platform for electrochemical detection (PCT/EP2016/050847):
- Control module: hardware/software that governs the microvalves and the regulation of the mycofluidic circuits.
- Loading module: allows the loading and mixing of fluids according to the specific recipe introduced by the user.
- Stirring module: by stirring the mixture with the different elements, the sample preparation process is accelerated.
- Measurement module: the functionalised magnetic particles are directed to the measurement area where the electrode is located, and once magnetically captured, the measurement is carried out.
SERVICES OFFERED BY THE ASSET
Analysis of idea / product
Initial analysis of the feasibility/opportunity of the idea compared to the state of the art.
Collaboration and co-working (generation of R&D proposals at regional, national and European level)
Search for potential partners for a collaborative proposal.
Pre-clinical validation
Realisation of prototypes for validation in relevant environments.
Proof of concept
Realisation of minimum viable products to validate the functionality of key technologies.
Strategic advice and support in the (co) development of new products or services (evaluation of ideas, specifications, early dialogue and health technology assessment, etc.)
Elaboration of a detailed state of the art on the technologies involved including intellectual property aspects.
ENTITY MANAGING THE ASSET

Contact person:
Santos Merino
santos.merino@tekniker.es